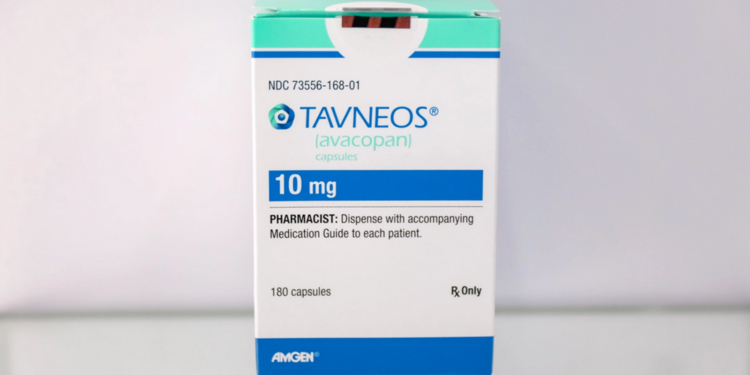
(MedPage Today) — The FDA on Tuesday warned that postmarketing data has turned up dozens of serious cases of drug-induced liver injury (DILI) associated with avacopan (Tavneos), a vasculitis drug that the agency wants removed from the market…
(MedPage Today) — The FDA on Tuesday warned that postmarketing data has turned up dozens of serious cases of drug-induced liver injury (DILI) associated with avacopan (Tavneos), a vasculitis drug that the agency wants removed from the market…
Source:
www.medpagetoday.com
Medical Disclaimer:
This content is shared for informational and educational purposes only.
It does not constitute medical, legal, or policy advice.
Healthcare decisions should always be made in consultation with qualified professionals.